Tag: biosimilars

How to Use the FDA Orange Book and Purple Book for Drug Safety Information
Learn how to use the FDA's Orange Book and Purple Book to identify withdrawn drugs, verify generic safety, and assess biosimilar risks. Essential for pharmacists, prescribers, and patient advocates.
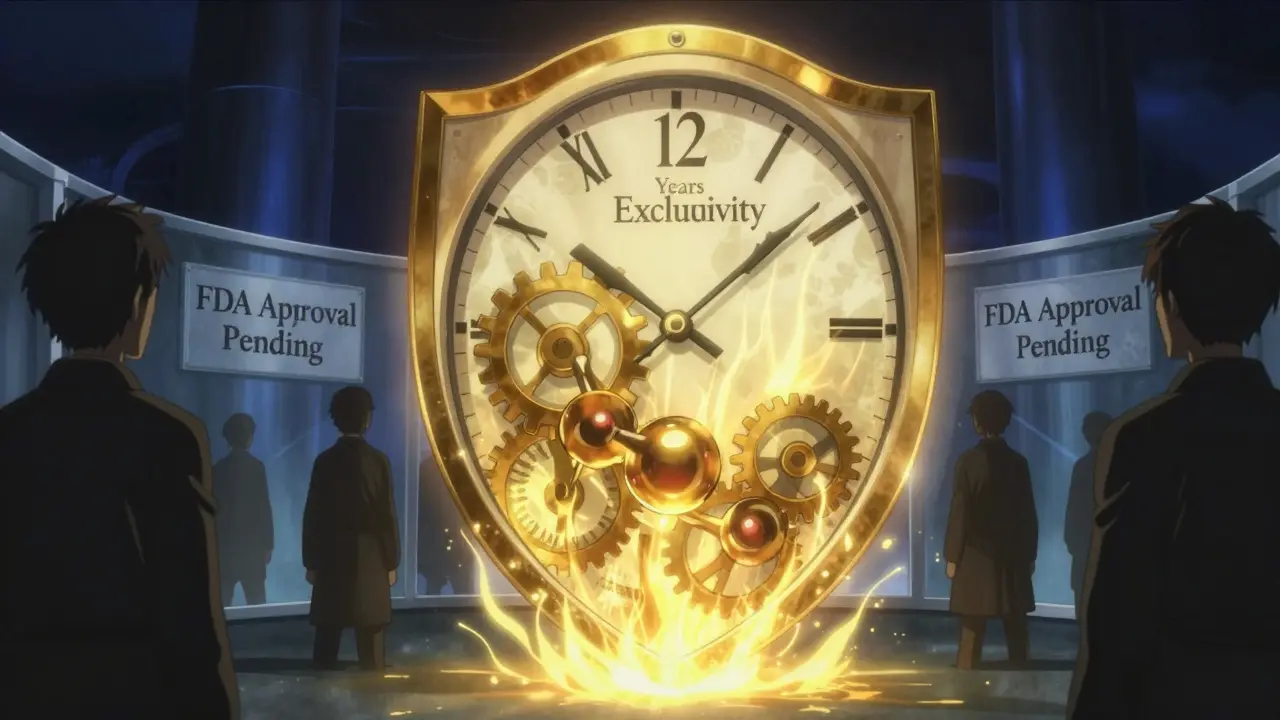
Biologic Patent Protection: When Biosimilars Can Enter the U.S. Market
Biosimilars in the U.S. face a 12-year exclusivity period before approval, delayed further by patent litigation and complex development costs. Unlike generics, they require massive investment and regulatory hurdles, leaving patients paying higher prices for years.
Categories
- Medications (95)
- Health and Wellness (47)
- Pharmacy Services (16)
- Chronic Conditions (9)
- Women Health (7)
- Health and Nutrition (5)
- Medical Research (4)
- Mental Health (4)
- Child Health (2)
- Skincare (2)


